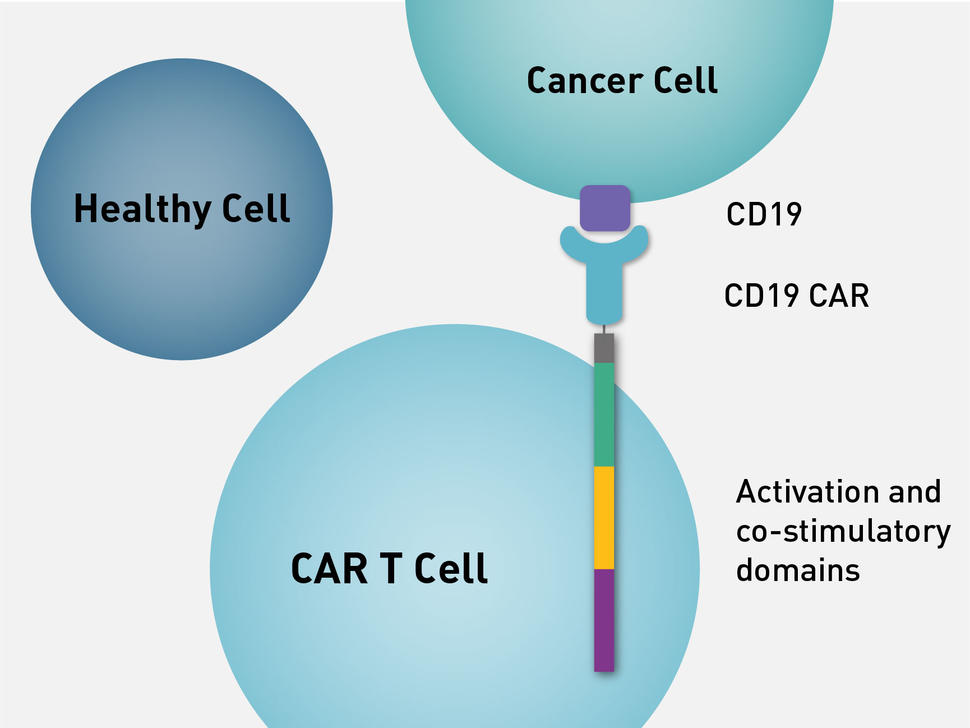
On November 8th, 2023, the National Medical Product Administration (NMPA) of China made an announcement that Inaticabtagene Autoleucel had been approved for the treatment of adults with relapsed or refractory B-cell acute lymphoblastic leukemia (r/r B-ALL). It is worth mentioning that this is also the first CAR-T product approved for the treatment of leukemia in China.